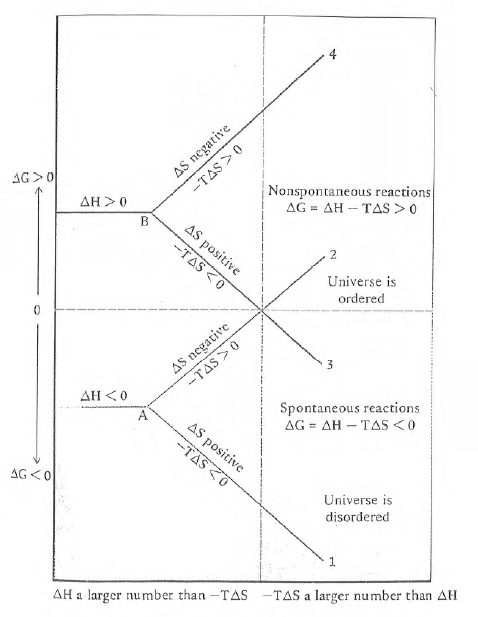
Figure 59. Changes in D G as a function of the
sign and magnitude of D S for an exothermic
reaction (D H < 0, point A) or an endothermic
reaction (D H > 0, point B). Point A corresponds
to disordering of the surroundings while point B corresponds to ordering
of the surroundings. When D S is negative,
the system is ordered and when D S is positive,
the system is disordered. The temperature dependence of the formal potential
of myoglobin uniquely follows line two and then abruptly follows
line three at 37C.
![]()