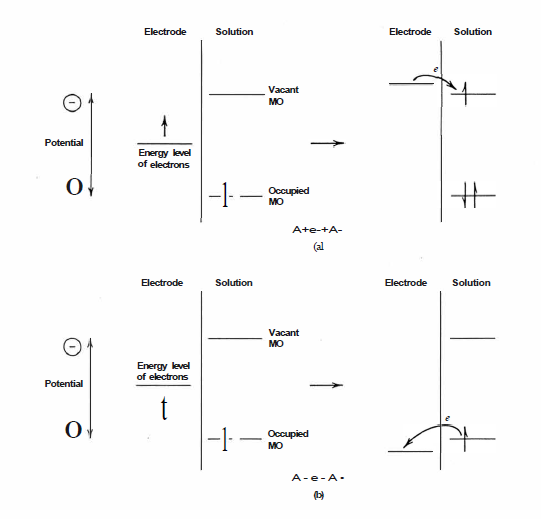
Figure 47. The value of E exhibited by an individual redox couple is a reflection of the relative stability of the reduced and oxidized states.
(a) Any factor which acts to stabilize the oxidized form makes the redox couple a better electron-donor and results in a more negative redox potential.
(b) Conversely, any factor which acts to stabilize the reduced form makes the couple a better electron-acceptor and gives rise to a more positive redox potential.